CHM 102: General Chemistry II
$ 23.99
Learning Track Courses

Introduction to Organic Chemistry (Undergraduate Foundation)
Carbon is the backbone of life and modern technology, yet most students struggle to visualise how its atoms connect. This course strips away the confusion surrounding organic chemistry by focusing on the fundamental rules that govern carbon behaviour. You will move from the basic history of the subject to the precise electronic arrangements that allow carbon to form millions of compounds. We cover hybridisation, bonding types, and molecular geometry with clarity. You will also explore the various forms of carbon, from diamond and graphite to advanced nanostructures like graphene and carbon nanotubes. This foundation is critical for understanding any advanced topic in chemistry or related sciences.
Understanding these concepts is not just about passing exams; it is about grasping the material world. Knowledge of bonding and polarity helps you predict how drugs interact with the body, how plastics are formed, and why certain materials conduct electricity while others do not. Engineers use these principles to design stronger composites; medical students need them to understand biochemistry; and industrial chemists rely on them to create new products. By mastering the structure of carbon allotropes and nanostructures, you gain insight into the technologies driving the future, from battery storage to nanomedicine. This knowledge turns abstract symbols into tangible tools for problem-solving in real-world scenarios.
By the end of this course, you will be able to determine the hybridisation state of any carbon atom and predict the three-dimensional shape of molecules. You will calculate formal charges, draw accurate resonance structures, and assess molecular polarity using electronegativity values. You will distinguish between the different allotropes of carbon and explain their unique physical properties based on their atomic arrangement. You will also understand the basic principles of nanochemistry and the significance of graphene and carbon nanotubes. These skills provide the necessary toolkit for tackling reaction mechanisms and spectroscopy in higher-level courses.
This course is built for first-year university students in chemistry, engineering, pharmacy, and medicine who need a solid start in organic chemistry. It is also ideal for advanced secondary school students preparing for university entrance exams or those who found their previous chemistry classes confusing. Even if you are not pursuing a science degree, this course offers a clear window into the molecular basis of life and technology. The explanations are direct and free of unnecessary jargon, making complex ideas accessible to anyone with a basic understanding of general chemistry. If you want to stop memorising and start understanding, this is the place to begin.
Introduction to Organic Chemistry (Undergraduate Foundation)
Carbon is the backbone of life and modern technology, yet most students struggle to visualise how its atoms connect. This course strips away the confusion surrounding organic chemistry by focusing on the fundamental rules that govern carbon behaviour. You will move from the basic history of the subject to the precise electronic arrangements that allow carbon to form millions of compounds. We cover hybridisation, bonding types, and molecular geometry with clarity. You will also explore the various forms of carbon, from diamond and graphite to advanced nanostructures like graphene and carbon nanotubes. This foundation is critical for understanding any advanced topic in chemistry or related sciences. Understanding these concepts is not just about passing exams; it is about grasping the material world. Knowledge of bonding and polarity helps you predict how drugs interact with the body, how plastics are formed, and why certain materials conduct electricity while others do not. Engineers use these principles to design stronger composites; medical students need them to understand biochemistry; and industrial chemists rely on them to create new products. By mastering the structure of carbon allotropes and nanostructures, you gain insight into the technologies driving the future, from battery storage to nanomedicine. This knowledge turns abstract symbols into tangible tools for problem-solving in real-world scenarios. By the end of this course, you will be able to determine the hybridisation state of any carbon atom and predict the three-dimensional shape of molecules. You will calculate formal charges, draw accurate resonance structures, and assess molecular polarity using electronegativity values. You will distinguish between the different allotropes of carbon and explain their unique physical properties based on their atomic arrangement. You will also understand the basic principles of nanochemistry and the significance of graphene and carbon nanotubes. These skills provide the necessary toolkit for tackling reaction mechanisms and spectroscopy in higher-level courses. This course is built for first-year university students in chemistry, engineering, pharmacy, and medicine who need a solid start in organic chemistry. It is also ideal for advanced secondary school students preparing for university entrance exams or those who found their previous chemistry classes confusing. Even if you are not pursuing a science degree, this course offers a clear window into the molecular basis of life and technology. The explanations are direct and free of unnecessary jargon, making complex ideas accessible to anyone with a basic understanding of general chemistry. If you want to stop memorising and start understanding, this is the place to begin.

Electronic Theory and Purification Techniques - Chemistry (Undergraduate Foundation)
Organic chemistry starts with how electrons move and hold atoms together. This course explains electronic theory, covering how bonds form and why electrons sit in specific places within a molecule. You will learn to isolate pure substances from mixtures using standard laboratory methods. We also cover how to identify what is inside a compound and calculate its exact chemical formula through qualitative and quantitative analysis.
Knowing how to purify chemicals is vital for making medicines, refining oil, and ensuring food safety. If you cannot separate a target substance from waste, you cannot manufacture a useful product. These techniques are the same ones used by forensic scientists to solve crimes and by engineers to develop new materials. Mastering these basics allows you to work accurately in any professional science lab or industrial setting.
By the end of this course, you will understand inductive effects, resonance, and how electron distribution dictates chemical reactions. You will be able to perform solvent extraction, chromatography, and distillation to get pure samples. You will also know how to test for specific elements like nitrogen or halogens and use mass data to determine empirical and molecular formulas. These skills ensure your laboratory results are reliable and scientifically sound.
This course is for first-year university students and science enthusiasts who want to move beyond textbook theory into practical lab work. It provides a solid base for anyone entering pharmacy, chemical engineering, or medical sciences. Even if you are just starting out, these lessons simplify complex ideas into clear steps, making it easier to grasp how modern science identifies the invisible components of the world around us.
Electronic Theory and Purification Techniques - Chemistry (Undergraduate Foundation)
Organic chemistry starts with how electrons move and hold atoms together. This course explains electronic theory, covering how bonds form and why electrons sit in specific places within a molecule. You will learn to isolate pure substances from mixtures using standard laboratory methods. We also cover how to identify what is inside a compound and calculate its exact chemical formula through qualitative and quantitative analysis. Knowing how to purify chemicals is vital for making medicines, refining oil, and ensuring food safety. If you cannot separate a target substance from waste, you cannot manufacture a useful product. These techniques are the same ones used by forensic scientists to solve crimes and by engineers to develop new materials. Mastering these basics allows you to work accurately in any professional science lab or industrial setting. By the end of this course, you will understand inductive effects, resonance, and how electron distribution dictates chemical reactions. You will be able to perform solvent extraction, chromatography, and distillation to get pure samples. You will also know how to test for specific elements like nitrogen or halogens and use mass data to determine empirical and molecular formulas. These skills ensure your laboratory results are reliable and scientifically sound. This course is for first-year university students and science enthusiasts who want to move beyond textbook theory into practical lab work. It provides a solid base for anyone entering pharmacy, chemical engineering, or medical sciences. Even if you are just starting out, these lessons simplify complex ideas into clear steps, making it easier to grasp how modern science identifies the invisible components of the world around us.
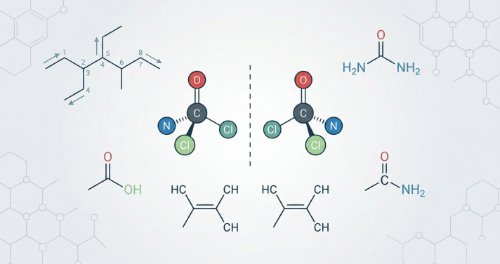
Organic Nomenclature and Stereochemistry (Undergraduate Foundation)
Chemical names follow strict laws. This course teaches IUPAC rules for naming organic compounds and how functional groups define their nature. You will learn to identify molecules by their structures and understand 3D spatial arrangements. We cover optical and geometric isomerism to show how identical formulas can have different shapes.
Doctors and pharmacists use these rules to identify drugs. In a lab, naming a substance wrongly can cause dangerous accidents. Knowing how atoms sit in space helps you understand how medicines fit into the human body. This knowledge is the base for making new materials, fuels, and life-saving treatments.
You will name complex organic molecules using standard IUPAC Systems. You will categorise compounds by their functional groups and predict their chemical behaviour. You will also identify different types of isomers and draw 3D structures accurately. By the end, you can explain how molecular shape affects biological function.
This course is for first-year university students in chemistry, pharmacy, and engineering. It also helps secondary school leavers preparing for advanced science exams. Even if you are not a science major, understanding these basics helps you read product labels and understand the chemicals in your daily life.
Organic Nomenclature and Stereochemistry (Undergraduate Foundation)
Chemical names follow strict laws. This course teaches IUPAC rules for naming organic compounds and how functional groups define their nature. You will learn to identify molecules by their structures and understand 3D spatial arrangements. We cover optical and geometric isomerism to show how identical formulas can have different shapes. Doctors and pharmacists use these rules to identify drugs. In a lab, naming a substance wrongly can cause dangerous accidents. Knowing how atoms sit in space helps you understand how medicines fit into the human body. This knowledge is the base for making new materials, fuels, and life-saving treatments. You will name complex organic molecules using standard IUPAC Systems. You will categorise compounds by their functional groups and predict their chemical behaviour. You will also identify different types of isomers and draw 3D structures accurately. By the end, you can explain how molecular shape affects biological function. This course is for first-year university students in chemistry, pharmacy, and engineering. It also helps secondary school leavers preparing for advanced science exams. Even if you are not a science major, understanding these basics helps you read product labels and understand the chemicals in your daily life.
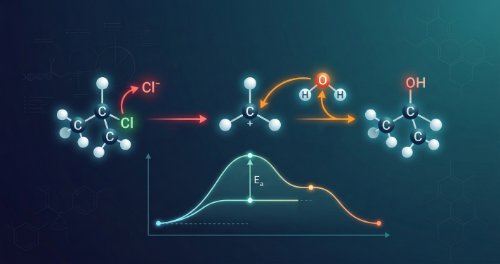
Organic Reaction Mechanisms and Kinetics - Chemistry (Undergraduate Foundation)
Organic reactions follow specific pathways called mechanisms rather than happening by chance. This course covers how molecules break and form bonds through step-by-step pathways and measured reaction rates. You will learn to track electron movement, identify intermediates, and understand the kinetics governing reaction speed. Studying these mechanisms allows you to predict how organic systems behave using fundamental chemical principles instead of simple memorisation.
Understanding these mechanisms is vital for industries like pharmacy, oil and gas, and food processing. Knowing how a reaction works allows you to control it to produce better medicines or stronger materials. These skills apply directly to laboratory research, chemical manufacturing, and quality control. Mastering reaction kinetics enables the optimisation of industrial processes, which saves time and reduces waste in commercial production.
You will gain the ability to draw reaction mechanisms using curly arrows to show electron flow and calculate reaction rates. The course teaches how temperature and catalysts change reaction speeds and helps you predict the final products of organic reactions. You will explain why specific pathways are preferred over others and master the relationship between energy changes and reaction timing in organic synthesis.
This course is for undergraduate chemistry and engineering students needing a solid foundation in organic chemistry. It also suits science teachers or laboratory assistants wanting to refresh their knowledge of reaction pathways. Even those new to the subject will benefit from clear explanations of bond-breaking and bond-making as a core part of chemical science. Any student preparing for advanced exams will find these logical steps useful for solving complex chemical problems.
Organic Reaction Mechanisms and Kinetics - Chemistry (Undergraduate Foundation)
Organic reactions follow specific pathways called mechanisms rather than happening by chance. This course covers how molecules break and form bonds through step-by-step pathways and measured reaction rates. You will learn to track electron movement, identify intermediates, and understand the kinetics governing reaction speed. Studying these mechanisms allows you to predict how organic systems behave using fundamental chemical principles instead of simple memorisation. Understanding these mechanisms is vital for industries like pharmacy, oil and gas, and food processing. Knowing how a reaction works allows you to control it to produce better medicines or stronger materials. These skills apply directly to laboratory research, chemical manufacturing, and quality control. Mastering reaction kinetics enables the optimisation of industrial processes, which saves time and reduces waste in commercial production. You will gain the ability to draw reaction mechanisms using curly arrows to show electron flow and calculate reaction rates. The course teaches how temperature and catalysts change reaction speeds and helps you predict the final products of organic reactions. You will explain why specific pathways are preferred over others and master the relationship between energy changes and reaction timing in organic synthesis. This course is for undergraduate chemistry and engineering students needing a solid foundation in organic chemistry. It also suits science teachers or laboratory assistants wanting to refresh their knowledge of reaction pathways. Even those new to the subject will benefit from clear explanations of bond-breaking and bond-making as a core part of chemical science. Any student preparing for advanced exams will find these logical steps useful for solving complex chemical problems.
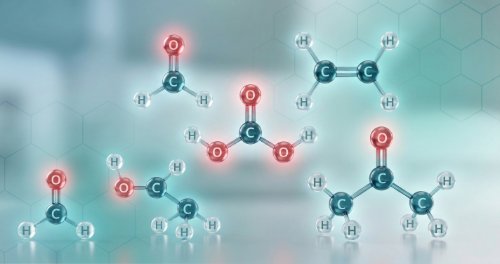
Chemistry of Functional Groups (Undergraduate Foundation)
Organic chemistry is the study of carbon compounds, and functional groups are the specific clusters of atoms that decide how these molecules behave. This course focuses on the properties and reactions of alkanes, alkenes, and alkynes, alongside the chemistry of alcohols, ethers, amines, and alkyl halides. You will also study the structure and reactivity of nitriles, aldehydes, ketones, and carboxylic acids to understand how millions of different molecules are organised into manageable families.
Understanding these groups is essential for producing everyday items like medicines, plastics, fuels, and soaps. This knowledge allows you to predict how chemicals will react in a lab, a factory, or inside the human body, making it vital for anyone pursuing a career in pharmacy, chemical engineering, or medicine. You will learn to identify these reactive parts and use them to solve practical problems in manufacturing and research.
By the end of this course, you will be able to name organic compounds using standard rules and predict the products of their chemical reactions. You will gain the skill to distinguish between different families of compounds based on their chemical tests and physical properties. You will also understand the mechanisms behind how these molecules transform into new substances, providing a solid foundation for advanced laboratory work and industrial applications.
This course is built for undergraduate students starting their journey in chemistry or related sciences like biochemistry and engineering. It is also useful for secondary school leavers preparing for university entrance exams who want a head start on complex topics. Even professionals in the health and manufacturing sectors will find this a useful refresher to better understand the chemical basis of the products they handle daily.
Chemistry of Functional Groups (Undergraduate Foundation)
Organic chemistry is the study of carbon compounds, and functional groups are the specific clusters of atoms that decide how these molecules behave. This course focuses on the properties and reactions of alkanes, alkenes, and alkynes, alongside the chemistry of alcohols, ethers, amines, and alkyl halides. You will also study the structure and reactivity of nitriles, aldehydes, ketones, and carboxylic acids to understand how millions of different molecules are organised into manageable families. Understanding these groups is essential for producing everyday items like medicines, plastics, fuels, and soaps. This knowledge allows you to predict how chemicals will react in a lab, a factory, or inside the human body, making it vital for anyone pursuing a career in pharmacy, chemical engineering, or medicine. You will learn to identify these reactive parts and use them to solve practical problems in manufacturing and research. By the end of this course, you will be able to name organic compounds using standard rules and predict the products of their chemical reactions. You will gain the skill to distinguish between different families of compounds based on their chemical tests and physical properties. You will also understand the mechanisms behind how these molecules transform into new substances, providing a solid foundation for advanced laboratory work and industrial applications. This course is built for undergraduate students starting their journey in chemistry or related sciences like biochemistry and engineering. It is also useful for secondary school leavers preparing for university entrance exams who want a head start on complex topics. Even professionals in the health and manufacturing sectors will find this a useful refresher to better understand the chemical basis of the products they handle daily.

Main Group and Transition Metal Chemistry (Undergraduate Foundation)
Periodic table mastery starts here. You will compare Group 1A, 2A, and 4A elements to see how their electron setups dictate chemical habits. The course connects these main groups to transition metals, focusing on how d-block metals form complex coordination compounds. This knowledge bridges the gap between organic and inorganic chemistry.
These principles drive industries from medicine to mining. You will understand how metals like iron and copper work in biological systems and industrial catalysts. Knowledge of coordination compounds is vital for purifying metals and creating new materials. This foundation helps you predict how substances react in a laboratory or a manufacturing plant.
You will learn to write electronic configurations for transition metals and explain the trends in Groups 1A, 2A, and 4A. You will identify the parts of coordination compounds, including ligands and central atoms, and calculate their oxidation states. By the end, you will be able to predict chemical properties based on an element's position in the periodic table.
This course suits undergraduate science and engineering students needing a solid chemistry base. It also helps secondary school graduates preparing for university entrance exams. Even those in related fields like pharmacy or metallurgy will find the clear breakdown of metal behaviour useful for their professional work.
Main Group and Transition Metal Chemistry (Undergraduate Foundation)
Periodic table mastery starts here. You will compare Group 1A, 2A, and 4A elements to see how their electron setups dictate chemical habits. The course connects these main groups to transition metals, focusing on how d-block metals form complex coordination compounds. This knowledge bridges the gap between organic and inorganic chemistry. These principles drive industries from medicine to mining. You will understand how metals like iron and copper work in biological systems and industrial catalysts. Knowledge of coordination compounds is vital for purifying metals and creating new materials. This foundation helps you predict how substances react in a laboratory or a manufacturing plant. You will learn to write electronic configurations for transition metals and explain the trends in Groups 1A, 2A, and 4A. You will identify the parts of coordination compounds, including ligands and central atoms, and calculate their oxidation states. By the end, you will be able to predict chemical properties based on an element's position in the periodic table. This course suits undergraduate science and engineering students needing a solid chemistry base. It also helps secondary school graduates preparing for university entrance exams. Even those in related fields like pharmacy or metallurgy will find the clear breakdown of metal behaviour useful for their professional work.